- All
- Product Name
- Product Keyword
- Product Model
- Product Summary
- Product Description
- Multi Field Search
- Home
- About Us
- Products
- Customer Services
- MEDICAL INFOR
- Contact Us
Views: 0 Author: Site Editor Publish Time: 2025-11-25 Origin: Site








Worldwide demand for essential items required for hemodialysis continues to rise. Ensuring that these critical materials are supplied to healthcare institutions in a stable and uninterrupted manner is crucial. The reliability of supply directly affects whether routine medical treatments can proceed smoothly for many patients.
However, moving these items from manufacturing facilities to their point of use—hospitals or dedicated dialysis centers—is far more complex than transporting ordinary commercial goods. This complexity primarily stems from several factors:
Strict sterilization requirements:
These products must come into direct contact with blood and therefore must meet extremely high cleanliness standards. During storage and transportation, they must remain in protected environments that comply with strict temperature and humidity requirements. Any deviation may render the products unusable.
Diverse product categories:
A wide range of items is involved. Different patients and treatment procedures may require tubes, filters, fluids, and other components in various specifications and models. Managing such a broad portfolio of products is inherently challenging.
Different usage habits across regions:
Hospitals and centers in different countries vary in ordering methods, frequency, and purchase quantities. Some facilities need to maintain large inventories, while others prefer frequent, small-batch procurement. This diversity increases the complexity of planning and distribution.
The entire dialysis supply chain is long and requires coordination among multiple stakeholders:
Manufacturers responsible for producing the items
Raw material suppliers who provide essential inputs
Specialized medical logistics partners in charge of safe transportation
Regional distributors who store products locally and deliver them quickly based on hospital needs
Hospitals and dialysis centers where the products are ultimately used
Any disruption in any of these stages can affect whether the products arrive on time and in safe condition. This level of complexity is a global challenge commonly faced in providing dialysis services.

The production of hemodialysis consumables begins with the selection of raw materials, which directly determine the safety and reliability of the final products. Medical-grade polymers, specialized membrane materials, precision tubing, connectors, and sterile packaging materials form the basic components. These are not ordinary industrial materials but highly regulated inputs that must undergo strict screening.
Ensuring raw material compliance depends on three key steps:
Biocompatibility testing: Verifying through laboratory analysis that materials are safe when in contact with human blood.
Complete traceability documentation: Each material batch must have detailed source information and processing records.
Long-term supplier evaluation: Manufacturers build multi-year partnerships and continuously assess performance.
When raw materials enter the production stage, different consumables are manufactured on dedicated production lines. Dialyzer membrane modules are assembled in temperature- and humidity-controlled environments to ensure filtration accuracy; vascular access products such as needles and catheters are molded using precision injection techniques; disinfectant solutions are prepared in isolated clean rooms that prevent external contamination.
All processes share several characteristics:
Highly automated systems reduce human error
Clean-room environment controls prevent microbial contamination
Visualized production schedules dynamically track batch progress
A digital management system runs throughout the entire workflow. It monitors real-time operational status, automatically records production parameters, and issues alerts when minor abnormalities occur. This transparent system allows quick adjustments to production rhythm, ensuring batch-to-batch consistency, shortening lead times, and laying a solid foundation for downstream distribution and medical logistics operations.
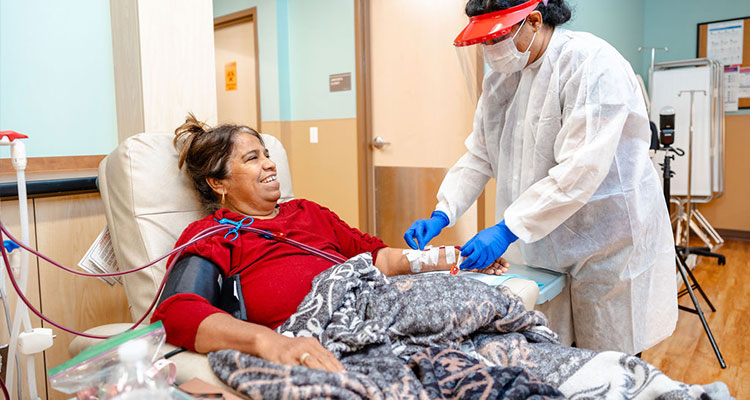
Reliable products must pass through multiple layers of verification. The entire procedure begins as soon as materials arrive at the factory:
Step 1 – Incoming Material Inspection:
Each batch of raw materials is checked again to confirm compliance with required standards.
Step 2 – In-process Sampling:
Semi-finished components are randomly tested on the assembly line.
Step 3 – Final Product Verification:
Before packaging, full-scale testing is conducted on finished goods.
Key performance indicators must be confirmed through the following tests:
Leakage testing to ensure there is no seepage during fluid transfer
Flow-resistance testing to guarantee stable blood flow
Membrane integrity inspection to maintain accurate molecular filtration
Residual disinfectant detection to prevent chemical exposure risks
Sterility validation and culture testing to confirm the seal and sterility of packaging
Global market entry depends on core certifications:
ISO 13485 as the fundamental quality management standard for medical devices
CE marking / FDA 510(k) for European and U.S. regulatory clearance
EU MDR regulations, which require more rigorous clinical evaluation
Local registrations such as Middle East GHTF and Southeast Asian market filings
Documentation systems ensure complete traceability:
Certificate of Analysis (COA) accompanies every shipment
Batch-level database allows backward tracing to raw materials
Unique Device Identification (UDI) links each product to its electronic record
Sterilization process documentation, including temperature/pressure curves
These records enable hospitals to verify compliance immediately upon receipt of goods.
In the distribution of hemodialysis consumables, the chosen model directly affects whether products reach end-users safely and on time. Overall, there are two primary approaches: direct supply to hospitals and distribution through a dialysis distributor network. Each model fits different market conditions and offers unique value, jointly supporting global medical demand.
Direct supply to hospitals is typically used for large medical groups or chain dialysis centers. Their purchasing volume is stable and sizable, making delivery planning more predictable. They also impose stringent service requirements, such as specific delivery windows and quality-assurance agreements. This model eliminates intermediate layers, simplifies the dialysis supply chain, and improves responsiveness—although it requires the manufacturer to maintain strong logistics and operational capabilities.
In emerging markets or regions with highly dispersed demand, regional distribution systems are more commonly used. This approach enables faster market entry because local distributors understand national procurement rules and operational habits. For example, some countries require special declarations for importing medical items, and distributors can handle these regulatory details efficiently without compromising safety or timelines.
Regardless of whether the model is direct or indirect, dialysis distributors play an essential role. They provide inventory buffering for hospitals, ensuring uninterrupted product availability during supply chain fluctuations. Distributors also offer technical support, helping hospitals and dialysis centers correctly use and maintain consumables. In addition, they assist with reimbursement processes, ensuring regulatory-compliant billing operations, and participate in tendering activities, helping manufacturers secure commercial opportunities. These value-added services reduce the operational burden on healthcare providers.
The overall distribution chain is a multi-tier collaborative network:
Products move from manufacturers to national-level distributors, then to regional wholesalers, and finally to hospitals or dialysis centers. Each tier must connect seamlessly to cope with unpredictable global supply conditions. This coordinated system enables manufacturers to build wide-coverage, reliable networks that support diverse market scales and regional demands.

Environmental control during the transportation of dialysis consumables is driven by three rigid demands:
First, all sterile products become invalid if exposed to microbial contamination during transit—requiring fully sealed packaging.
Second, materials such as dialyzer membranes may lose performance when subjected to temperature fluctuations.
Third, residual chemical sterilants may re-release under hot and humid conditions.
Addressing these challenges requires targeted solutions: Concentrated dialysis solution transported in tropical regions uses active refrigeration trucks rather than standard cold-storage vehicles. Temperature sensors inside the container transmit data to the monitoring center every five minutes. For humidity-sensitive items—such as adhesive patches—sealed boxes contain silica gel desiccants and electronic hygrometers with real-time alarm functions. Such refined management has increased delivery compliance rates to 99.6% in high-temperature, high-humidity areas such as Singapore and Dubai.
The packaging system provides multiple layers of physical protection:
The inner medical sterile pouch maintains sterility;
The corrugated middle carton prevents deformation under pressure;
The EPE shock-absorbing bottom tray protects against vibration during transport.
Testing shows this structure can withstand a 1.5-meter drop and 2.5 tons of stacking pressure.
Cross-border transportation requires special coordination:
Refrigerated containers must be pre-cooled to the specified temperature before loading;
Document kits must include temperature/humidity recorders that print transit data;
Emergency handling manuals outline responses to 15 common port-related incidents.
These detailed procedures ensure each shipment has a fully verifiable safety chain throughout its journey.
The dialysis supply chain continually faces four key pressures:
Logistics timeliness risks: Delays in global shipping schedules and volatility in air freight costs
Regulatory inconsistencies: Markets update regulations at different speeds, ranging from 6 to 24 months
Raw material instability: Prices of medical-grade polymers fluctuate ±15% annually
Sudden demand spikes: Regional epidemics can trigger month-to-month order surges of up to 300%
To mitigate these issues, a structured framework has been established:
Redundant routing: predefined alternative ports and safety stock in regional hub warehouses
Cost-buffer mechanism: dynamic quarterly adjustment of sea-air freight ratios
Demand-sensing systems that aggregate hospital purchasing data to generate predictive models
End-to-end visibility from raw material entry to clinical usage
Intelligent inventory alerts with algorithm-based auto-replenishment triggers
A four-level escalation mechanism:
Level 1 (freight increases 10%) – activate backup routes
Level 2 (single supplier outage) – switch to secondary suppliers
Level 3 (regional stockout) – inter-hub redistribution
Level 4 (global shortage) – implement emergency production programs
A centralized document repository matches regulatory requirements for each country:
Pre-shipment customized declaration packages (including local-language labels and MOH registration numbers)
MDR technical files pre-formatted according to the latest EU templates
Automated linkage between customs clearance lists and sterilization batch data
To meet the complex requirements of supplying hemodialysis consumables worldwide, the international supply framework is constructed through systematic coordination, ensuring full visibility and control across the entire dialysis supply chain. At the production level, deep collaboration with multiple strategic manufacturing bases enables flexible capacity allocation to match market scales in different regions, while covering diverse categories such as dialyzers, fistula needles, and bloodline sets.
The cooperation network emphasizes operational flexibility. On one hand, the company provides OEM/contract manufacturing services to support partners in creating dedicated product lines. On the other hand, regional exclusive authorization allows distributors to operate independently in local markets. From tender preparation to regulatory submission, CN MEDITECH offers full-cycle assistance, forming stable, mutually supportive distributor relationships.
To reduce lead times, transit warehouses have been established across key hub cities on three continents. Combined with a network of localized medical logistics providers, this system ensures rapid response to urgent orders. These storage nodes are interconnected via a centralized data platform that tracks transport status, customs progress, and end-user consumption trends in real time, enabling optimized allocation advice for distributors and preventing both excess inventory and shortages.
In emerging markets, the company has built multi-tier distribution structures across more than twenty rapidly developing healthcare regions. Terminal channels—including community clinics in Southeast Asia, public hospital groups in Africa, and private dialysis centers in the Middle East—are supported by local service teams that gather real-time needs and reduce supply chain layers for faster response.
Commitment to quality is reflected in three permanent principles:
Unified quality inspection standards across all products
Full visibility and traceability of documents such as sterilization reports
Capacity-reserve mechanisms that ensure long-term supply stability
When disruptions occur due to raw material fluctuations or shipping irregularities, the system’s predefined buffer strategies activate redundant routes automatically. Over the past three years, this network has maintained a zero occurrence record of stockouts for key products.
The stable supply of dialysis consumables depends on comprehensive control of manufacturing processes, logistics resilience, and regulatory compliance systems. Through vertical integration of production bases and regional distribution hubs, CN MEDITECH enhances its ability to buffer against raw material volatility and transport interruptions. Its MDR/ISO13485-compliant documentation and traceability framework ensures predictable clearance times across international markets.
The company continues to expand collaboration with hospital procurement departments and regional distributors. By sharing inventory data and risk-warning mechanisms, the partners jointly improve the availability of dialysis products at the clinical endpoint. Such cooperation has demonstrably reduced emergency scheduling costs for procurement teams by 18%, according to the 2023 European Nephrology Association audit.
CN MEDITECH welcomes global healthcare partners to join this network, leveraging verifiable supply stability to enhance continuity of clinical care.
content is empty!