- All
- Product Name
- Product Keyword
- Product Model
- Product Summary
- Product Description
- Multi Field Search
- Home
- About Us
- Products
- Customer Services
- MEDICAL INFOR
- Contact Us
Views: 0 Author: Site Editor Publish Time: 2025-10-23 Origin: Site








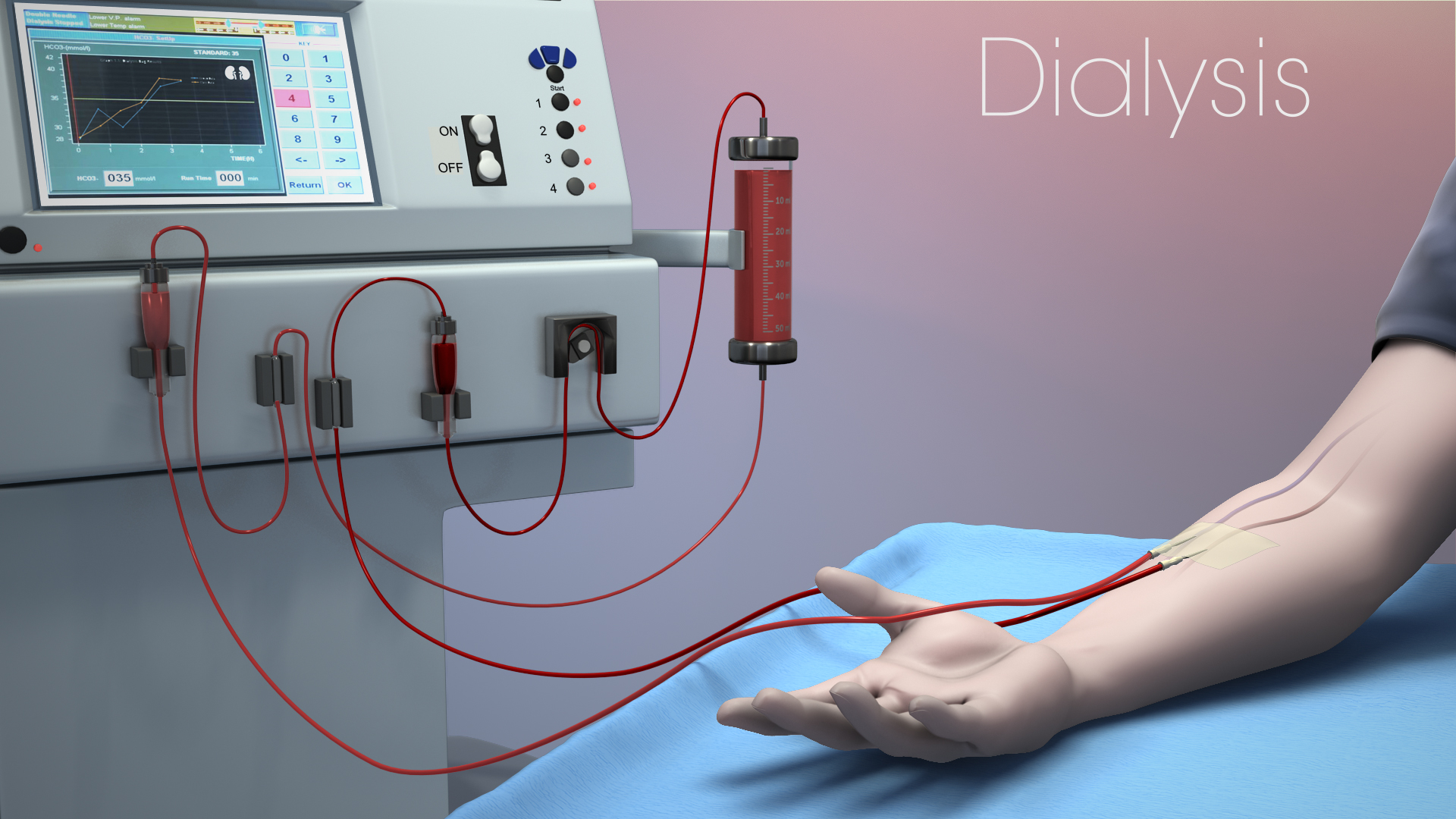
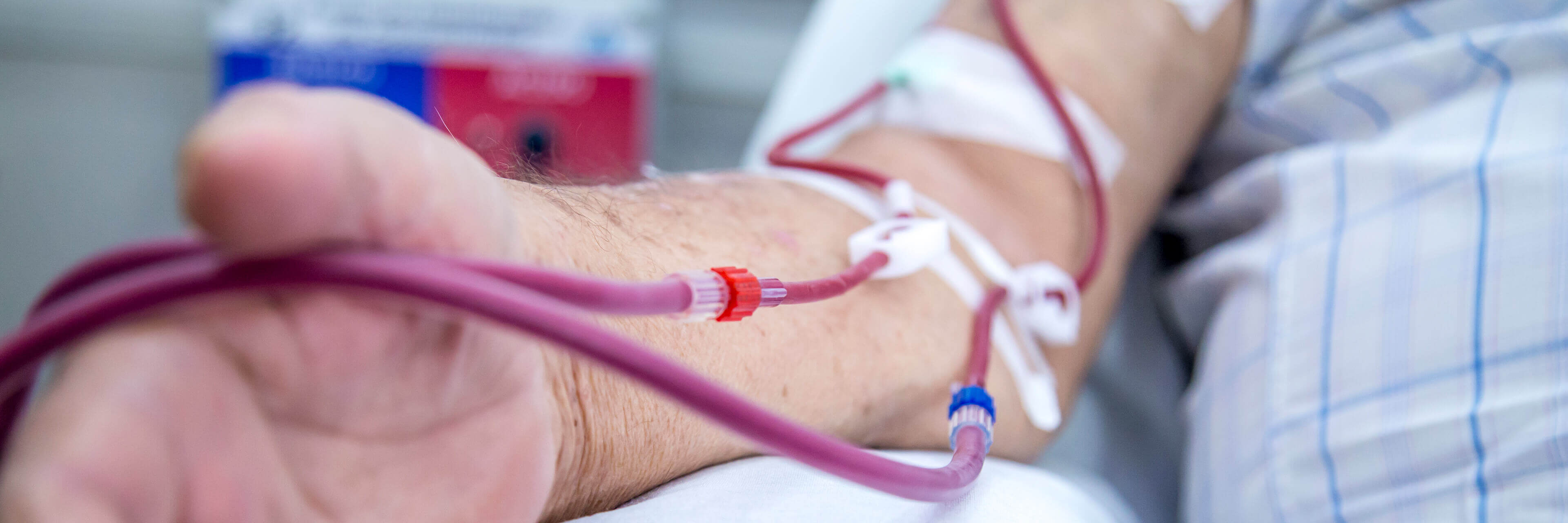
In hemodialysis treatment, patient safety always comes first. One of the most critical aspects of ensuring safety is preventing infection during treatment. Using disposable hemodialysis supplies—such as dialyzers, tubing sets, and fistula needles—is the most basic and effective way to build this essential safety barrier.
Each hemodialysis session involves direct contact with the patient’s blood system. If supplies are reused—even after strict cleaning and disinfection—there remains a small risk of residual contaminants or microorganisms spreading between patients. Disposable supplies, strictly following the “one patient, one use, one discard” principle, completely eliminate this hidden danger. They provide every patient with a brand-new, fully independent treatment pathway, minimizing the possibility of bloodborne or fluid-borne pathogens transferring from one patient to another.
Major international health authorities, such as the World Health Organization (WHO) and the U.S. Centers for Disease Control and Prevention (CDC), explicitly recommend the use of disposable medical supplies in their infection control guidelines as best practice. These guidelines clearly emphasize that single-use medical devices are a key operational measure to reduce the transmission of bloodborne pathogens. Therefore, selecting disposable products that comply with these standards demonstrates responsibility for the overall safety of the dialysis treatment process.
Compared to reusable dialysis components, disposable consumables undergo final sterilization and quality inspection before leaving the factory. During clinical use, this eliminates the complicated and error-prone steps of cleaning, disinfection, and verification. Every oversight in those processes could introduce new risks. Disposable items are ready to use straight from the package, ensuring consistent and stable quality while significantly reducing the risk of adverse outcomes caused by incomplete cleaning or sterilization.
It is especially important to select disposable hemodialysis consumables approved by authoritative organizations such as CE (Europe) and FDA (United States). These certifications mean that the product’s design, manufacturing process, performance verification, and sterilization have passed stringent regulatory evaluation—proving their clinical safety. Using such certified products brings higher confidence for both healthcare professionals and patients and directly supports the clinical goal of achieving lower cross-contamination and infection rates.
This is the core component of dialysis treatment. Its internal membrane structure efficiently removes excess solutes and fluids from the patient’s blood. Various models are available with different filtration capacities.
The models we provide have core filtration membranes (material type) and overall designs that have been strictly evaluated and certified under CE or FDA standards, ensuring both safety and efficacy. This includes verification of filtration performance, sterilization process integrity, and patient comfort (biocompatibility).

These are essential tubes that connect the patient, dialyzer, and dialysis machine, forming the extracorporeal blood circulation circuit.
The tubing system complies with internationally recognized safety standards (such as ISO 8637). The connection joints are precisely designed to prevent leakage. All materials are certified biocompatible and free from harmful substances. The final sterilization process (e.g., ethylene oxide sterilization) and its validation strictly conform to CE/FDA requirements.
Used for preparing the dialysate solution, including acid concentrates, bicarbonate powders, or ready-to-use liquids.
Composition and microbial limits strictly comply with relevant international standards (such as ISO 23500). All export-oriented product batches carry CE marking with traceable identification. Packaging labels (in multiple languages) ensure accurate and transparent information delivery.

Specialized needles designed to connect to the patient’s vascular access during hemodialysis.
Smooth surface treatment (e.g., silicone coating) ensures patient comfort. Winged design facilitates stable fixation during use. Needle sizes are available in multiple options with clear color-coded identification. The design and manufacturing of all models undergo CE or FDA certification to guarantee safety in long-term clinical use.

These are preassembled sterile kits containing essential tools (catheter, guidewire, scalpel, syringe, etc.) and accessories needed for placing a dialysis catheter.
The fully sterile packaging allows healthcare professionals to use the kit directly, enabling faster and safer establishment of temporary or permanent vascular access. Every sterilized item in the kit is validated for compliance with CE/FDA sterilization requirements.
Also known as “B Powder Canisters” or “B Powder Bags,” these contain dry bicarbonate powder used by dialysis machines for real-time dialysate preparation.
Compared to large-volume liquid concentrates, they are smaller, lighter, and significantly reduce transportation cost and complexity while minimizing storage space requirements. They are easy to use and have a low contamination risk. Certified products meet regulatory standards for dissolution performance and safety inside the dialysis machine.
The CE mark serves as the mandatory “passport” for products entering the European Economic Area (EEA). It indicates that the product fully complies with the EU’s strict requirements for safety, health, and environmental protection.
Obtaining CE certification means that both our production system (based on the ISO 13485 Quality Management System) and every individual product have undergone in-depth audits and evaluations by EU-designated notified bodies. The manufacturer bears the crucial responsibility of maintaining continuous product compliance after certification.
This is one of the key regulatory procedures by which the U.S. Food and Drug Administration (FDA) authorizes the marketing of medical devices in the United States. It ensures that the device has been proven to be substantially equivalent (no less safe or effective) to a legally marketed predicate device.
To obtain this clearance, manufacturers must submit detailed documentation to the FDA, including product descriptions, performance data (such as mechanical testing, biocompatibility, and sterilization validation), demonstrating safety and effectiveness. Achieving FDA clearance represents formal recognition from U.S. regulatory authorities.
ISO standards are globally recognized technical frameworks developed by the International Organization for Standardization. They define consistent and rigorous benchmarks for specific domains, including production quality systems, product performance, and safety requirements.
· ISO 13485:
This is not a product certification but an international standard for quality management systems specific to medical devices. It forms the foundation for achieving CE certification and FDA GMP compliance, ensuring production processes remain controlled, stable, and continuously meet product quality requirements.
· ISO 23500 / ISO 8637:
These standards directly address the performance and safety of dialysis-related materials, such as dialysate composition and purity, and the safety requirements for extracorporeal bloodline tubing systems.
To ensure smooth market access globally (especially in non-EU and non-U.S. regions), certain official supporting documents are often required:
· Certificate of Free Sale (CFS):
Issued by the national regulatory authority (such as the drug or medical device administration in the manufacturing country), this document certifies that the product is legally allowed for sale in its domestic market—often a prerequisite for customs clearance or product registration abroad.
· Declaration of Conformity (DoC):
Issued by the manufacturer, this formal document declares that the product complies with specific regulatory frameworks (such as EU MDR).
· Market Access Guarantee: These certifications act as regulatory passports, enabling your products to cross international compliance barriers and enter target markets legally.
· Reliable Proof of Quality: They serve as objective, verifiable evidence of product quality and adherence to international standards—removing the need for exaggerated marketing claims.
· Risk Reduction: Selling products with comprehensive certification and documentation significantly minimizes the risk of reputational or financial losses caused by non-compliance or product quality incidents.
When selecting a manufacturer partnership for hemodialysis consumables worldwide, ensuring that the partner has a robust and well-managed quality system is the foundation of a successful collaboration. The following key elements form a practical framework for evaluating supplier reliability and establishing a trustworthy working relationship—not merely a service checklist.
Before committing to large-scale procurement, always insist on providing and signing a detailed sample evaluation agreement.
This document should clearly specify:
- The number and specifications of samples available for testing;
- The key testing items (such as sterility validation, performance tests including flow rate and sealing integrity).
This allows you to verify whether the product’s key performance indicators meet your target market’s requirements or align with industry-accepted standards—while maintaining controlled risk through small-volume trial orders.
Request the manufacturer to demonstrate and implement a strict batch management system.
All products and primary packaging must bear unique, clearly identifiable lot numbers/serial numbers and expiry dates.
Confirm the existence of complete traceability records to ensure that the manufacturer can promptly provide corresponding production, process control, and final release testing documentation (with marked “pass” results).
Also, establish mutual access rights and document retention periods.
Such traceability enables efficient post-market activities—such as quality investigations, complaint handling, or recalls if necessary—fulfilling market surveillance responsibilities and protecting brand reputation.
Verify the cleanroom compliance of the production facility.
Manufacturers should provide documentation showing that their key production areas (e.g., purified filling, assembly) meet the cleanroom classification defined in ISO 14644, along with valid environmental monitoring records.
Understand process transparency by clarifying key quality control points in core manufacturing steps (such as sealing, welding, and in-line inspection) and the corresponding methods (automatic, manual, sampling frequency).
This proactive verification reduces risks of contamination or process variation at the source and ensures consistent and reliable product quality—forming the physical foundation of clinical safety.
Insist on regularly updated biocompatibility and safety test reports, and review official reports issued by reputable, internationally accredited independent laboratories (such as GLP or ISO 17025 certified labs).
Pay attention to the scope and methodology of the tests. Confirm that key items—such as cytotoxicity, sensitization, and intracutaneous reactivity—cover all materials that come into contact with the human body and comply with globally accepted test standards.
These reports serve as objective evidence beyond the manufacturer’s self-assessment, providing authoritative support when demonstrating the product’s biological safety to regulatory agencies or end customers.
Suppliers must hold and maintain valid core certifications such as ISO 13485 quality management system certificates—these are non-negotiable entry requirements.
Make use of the manufacturer’s offered remote or virtual audit support, which may include:
· Real-time video sessions to inspect key workshops and production processes;
· Secure online access to core documents (e.g., quality manual, SOPs, change control records);
· Direct Q&A or review of critical quality system elements (such as CAPA, complaint handling, and deviation management).
Manufacturers should promptly provide genuine evidence of corrective actions taken in response to audit findings.
This approach allows cost-effective and efficient verification of whether the claimed quality system is not only in place but effectively implemented. It also helps establish transparent, fact-based communication channels, encouraging continuous improvement and collaborative problem-solving.
When engaging in a manufacturer partnership with Chinese suppliers in the hemodialysis field, you gain access to a combination of certified quality, strong production capability, and cost efficiency that is unmatched in many global markets.
Supported by mature industrial clusters and highly integrated upstream and downstream supply chains, we provide products that strictly comply with your required quality standards and international certifications (such as CE and FDA)—all while maintaining highly competitive pricing.
This cost-effectiveness is not achieved through “low prices,” but rather through value optimization—the result of large-scale production, efficient resource utilization, and localized supply chain maturity.
You gain long-term cost control advantages, enabling you to maintain healthy profit margins while remaining price-competitive in your markets—or reinvest the savings into marketing and business development.
We fully support your needs for private-label manufacturing (dialysis OEM) and joint design development (dialysis ODM), offering comprehensive customization capabilities:
· Brand Customization: Complete adaptation to your packaging design, brand logo, and labeling requirements—whether for retail distribution or hospital channels.
· Market Adaptation: Multi-language labeling and documentation tailored to meet regulatory and usage requirements in your target markets.
Rapid response to changing end-user preferences, enhanced brand identity, and strengthened differentiation in competitive niche markets.
Located within one of the world’s leading medical device manufacturing clusters, Chinese dialysis suppliers operate with strong production capacity and well-integrated global logistics systems. Their stable supply network spans over 80 countries, supported by close cooperation with top-tier international logistics providers.
This minimizes the risk of supply chain disruptions. Even under challenging external conditions (e.g., shipping delays or port congestion), you can still receive consistent product supply with shorter lead times—ensuring reliability for your downstream clients.
Optimized production processes, advanced scheduling systems, and efficient warehouse management significantly reduce turnaround time from raw material sourcing to finished product shipment.
With manufacturing hubs located close to major international seaports, large-volume orders can be cleared and shipped quickly.
Greatly reduced end-to-end lead times from order placement to delivery, improved inventory turnover efficiency, faster response to market fluctuations, and reduced working capital pressure.
Professional suppliers—such as CN MEDITECH, specializing in dialysis consumables—bring extensive experience and regulatory expertise to global collaborations:
· Certified Product Portfolio: A wide range of CE- and FDA-certified products, all holding valid export qualifications for medical device distribution.
· Ongoing Regulatory Monitoring: Dedicated compliance teams continuously track updates to major market regulations (Europe, U.S., Middle East, Latin America, Southeast Asia, etc.).
· Registration Support: Capability to provide technical documentation packages that meet target market requirements (without acting as a legal representative).
Significantly reduced global market entry barriers and time-to-market; improved registration efficiency; and the ability to leverage the supplier’s regulatory best practices to build a strong compliance foundation for your international operations.
Emerging markets are experiencing strong growth in demand for hemodialysis services, creating unique opportunities for international distributors with strategic vision.
Regions such as the Middle East (e.g., Saudi Arabia, UAE), Eastern Europe (Poland, Czech Republic), and Africa (Kenya, Nigeria) are witnessing a sharp rise in the prevalence of End-Stage Renal Disease (ESRD). Combined with expanding government healthcare investment, hospitals and dialysis centers are seeking reliable, certified dialysis consumable suppliers.
· Public Tenders: Closely monitor open tenders organized by national health insurance programs or large hospital groups. These projects typically require suppliers to hold valid CE/FDA certifications and comprehensive quality documentation.
· Private Sector Growth: The rapid expansion of privately owned dialysis centers has significantly increased demand for cost-effective, compliant, and high-quality consumables.
· Exclusive Regional Authorization: Build long-term trust through exclusive partnerships in specific countries or regions, working together to penetrate and develop local markets.
· Joint Tender Participation: Collaborate closely with your supplier to obtain complete sets of technical documents and qualification packages required for public tenders—enhancing your bidding success rate.
· Own-Brand (OBM) Solutions: Leverage the supplier’s dialysis OEM/ODM capabilities to quickly launch your own branded product line, establishing a strong localized market identity.
· Regional Hub Strategy:
Select strategic logistics hubs with regional coverage potential—such as Dubai (for Middle East & Africa) and Warsaw (for Eastern Europe)—as distribution centers.
By leveraging Chinese suppliers’ mature logistics networks, you can achieve rapid restocking, reduced inventory pressure, and shorter local delivery times.
Chinese dialysis manufacturers can provide:
· Pre-approved marketing materials, clinical data summaries, and full regulatory documentation packages (including CE/FDA certificates) that comply with tender requirements.
· Professional after-sales technical teams ready to assist with early-stage clinical usage inquiries or provide basic operational training on demand.
· Seamless access to their mature global export networks, ensuring stable supply to your regional distribution centers.
Seize the Momentum
The supply-demand gap for dialysis consumables in emerging markets is substantial—and early market entry provides a critical advantage.
By combining China’s strengths in compliance, cost-effectiveness, supply chain efficiency, and flexible cooperation models, you can unlock the potential of these high-growth regions efficiently, build strong market presence, and secure long-term revenue streams.
As a mature international trading company specializing in the field of hemodialysis, CN MEDITECH is dedicated to being your reliable and long-term manufacturer partnership.
Ø Strict Certification Ensures Trust: We carefully select product portfolios certified by CE and FDA, guaranteeing safe, compliant, and reliable dialysis consumable supplies.
Ø Global Resource Network Empowerment: Leveraging cross-regional supply systems and deep market experience, we help you efficiently expand into emerging markets while minimizing operational complexity.
Ø Agile Local Response: We provide customized supply solutions, tender support, and post-sales coordination—acting as your strategic support point in business expansion.
Contact us today to receive your exclusive regional cooperation proposal, certified product catalog, and complete export compliance documentation package.
Let’s seize the opportunity to grow together and capture the expanding global dialysis market.
→ Contact Email: meditech@cn-meditech.com / elvanenglish1984@gmail.com
content is empty!